Vitiligo
About Vitiligo
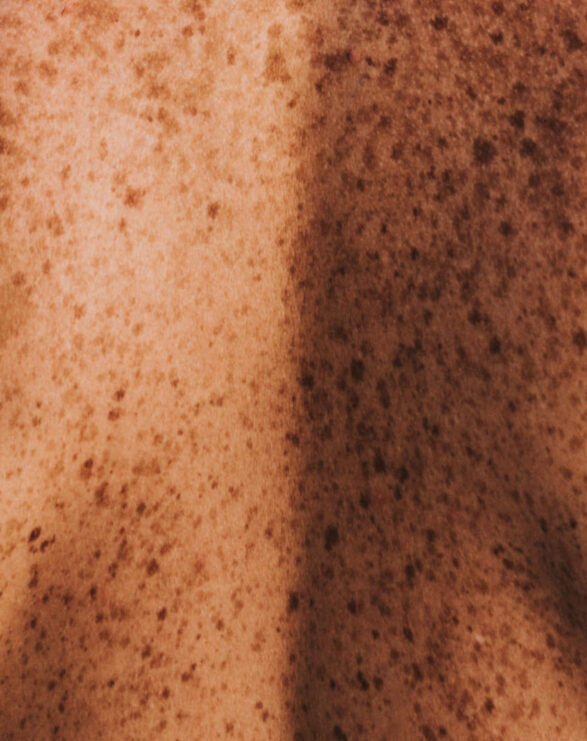
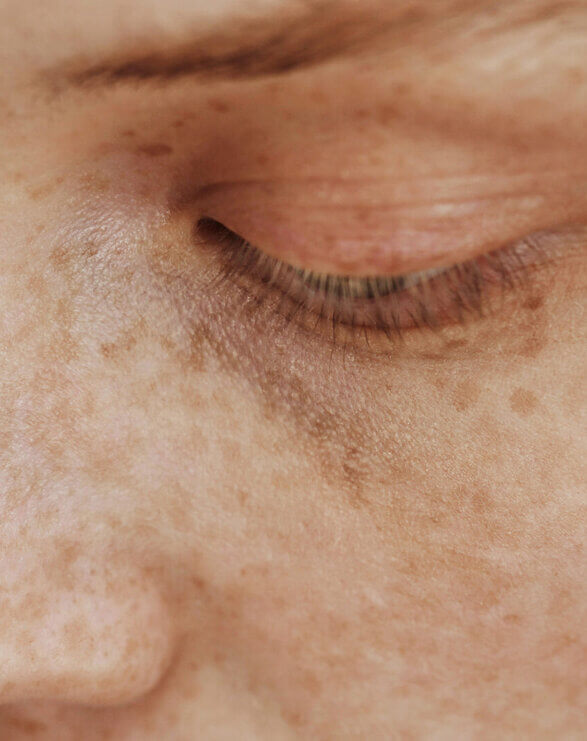
Vitiligo is a skin disorder in which the pigment-producing cells of the skin (melanocytes) are absent or demonstrate a lack of activity. As a result, lighter depigmented patches of skin (vitiliginous lesions) appear in different parts of the body due the lack of melanin (pigment). The exact cause of vitiligo is unknown, but it is generally recognised that an autoimmune component plays a role in this disease. Between 0.1-2% of the global population is affected by vitiligo impacting all races. Vitiligo causes significant psychological and emotional distress.
Vitiligo is traditionally separated into two clinical forms: generalised vitiligo and segmental vitiligo (SV), which present with distinctive clinical features and natural histories.
Generalised vitiligo (previously referred to as nonsegmental vitiligo)* is the most common form of the disease, accounting for 72-95% of cases. The vitiliginous lesions are usually symmetrically distributed and new patches may appear throughout the patient’s life. The disease is progressive with flare-ups. Vitiligo is frequently associated with personal or family history of autoimmunity.
SV is characterised by a unilateral distribution that may totally or partially match a dermatome (area of skin with innervation from a single spinal nerve) and has an earlier onset and a rapid spread. SV occurs in a minority of patients and is thought to be more frequent in pediatric patients; it may account for 30% of childhood cases. Autoimmune association is rare with SV.
The main goal of treating vitiligo is to achieve an arrest of the depigmentation or even arrive at repigmentation of the unpigmented lesions. Many treatment options exist but clinical challenges persist, as not all patients respond to available therapies and relapse is common.
CLINUVEL commenced a pilot Phase II trial of SCENESSE® (afamelanotide 16mg) as a repigmentation therapy in patients with generalised vitiligo in the USA and Europe in the second quarter of 2011. SCENESSE® is being evaluated for its ability to activate melanocytes within the vitiliginous lesions and achieve repigmentation in combination with NB-UVB in patients with vitiligo. The first treatment results from this programme, study CUV102, were announced in December 2012. In CUV103, a proof-of-concept study in Singapore, a more pronounced clinically meaningful recurrence of pigmentation for total body and areas of the head and neck was observed. In October 2023 CLINUVEL announced the start of a Phase III clinical trial, CUV105, evaluating SCENESSE® in vitiligo.
Understanding Vitiligo – Causes and Theories
Take a deeper dive into vitiligo – the possible pathogenesis of the disease and its effects.
Clinical & regulatory progress
January 2026
New clinical reports from four patients enrolled in CLINUVEL’s ongoing study of SCENESSE® (afamelanotide 16mg) in vitiligo (CUV105) were presented to the 31st Regional Dermatology Training Center (RDTC) Continuing Medical Education (CME) Conference in Moshi, Tanzania. Read more.
September 2025
New patient cases demonstrating the efficacy of SCENESSE® (afamelanotide 16mg) in vitiligo patients were presented to the European Academy of Dermatology and Venereology (EADV) conference in Paris, France. Read more.
June 2025
CLINUVEL released an extended global update on the vitiligo landscape and the Company’s vitiligo program. See the update.
May 2025
CLINUVEL announced that the Company has met its recruitment target in its Phase III trial (CUV105) of SCENESSE® (afamelanotide 16mg) in vitiligo, with more than 200 patients enrolled. Read the announcement.
January 2025
CLINUVEL shared four case reports from its ongoing CUV105 Phase III study in vitiligo. More details here.
March 2024
A new case on the clinical efficacy of SCENESSE® (afamelanotide 16mg) in vitiligo has been presented during a plenary session of the American Academy of Dermatology (AAD) meeting in San Diego on 9 March. More details here.
March 2024
Clinical data from the use of CLINUVEL’s drug SCENESSE® (afamelanotide 16mg) in porphyrias and vitiligo were presented at the American Academy of Dermatology (AAD) Meeting. Read the announcement.
October 2023
CLINUVEL announced that SCENESSE® (afamelanotide 16mg) has been administered as a systemic treatment to the first vitiligo patient in a Phase III study, CUV105. Up to 200 adolescent and adult vitiligo patients with darker skin types (Fitzpatrick Types IV-VI) will be enrolled in the study, comparing the use of SCENESSE® in combination with narrowband UVB (NB-UVB) light therapy to NB-UVB as a monotherapy. Read the announcement.
September 2023
CLINUVEL enters a formal collaboration with four of the world’s leading vitiligo experts, who will support clinical and scientific work and provide advice on the Company’s late-stage development of, and path to market for, SCENESSE® (afamelanotide). Read the announcement for details on the Vitiligo Expert Panel.
October 2022
The first vitiligo patient has been enroled in the CUV104 study. Read the announcement.
May 2022
CLINUVEL receives positive review from the Institutional Review Board (IRB) to commence the CUV104 study, which will be conducted at a North-American vitiligo expert centre. Read the announcement.
December 2021
FDA agrees to Phase II clinical protocol evaluating SCENESSE® (afamelanotide 16mg) as a monotherapy in patients with vitiligo (CUV104 study). The study will commence at a specialist hospital department in the USA. Read the announcement.
May 2020
CLINUVEL meets the US FDA and global experts to discuss the North American development program for the treatment of vitiligo. Read this announcement.
December 2018
CUV103 Singapore Phase II vitiligo study results released.
December 2015
North American program update published.
May 2014
Further Phase II study commenced in Singapore with preliminary results reported in December 2015.
2012
Updated clinical observations presented alongside 2012 AAD Meeting and observations from US vitiligo study published. First treatment results announced: SCENESSE® successful in Phase IIa vitiligo study.
2011
Trial allowed by US Food and Drug Administration (FDA); Pilot commenced; and first clinical observations presented at 2011 EADV congress.
August 2010
Programme announced.
Join our Vitiligo newsletter
References
Toh JJ, Chuah SY, Jhingan A, Chong WS, Thng ST. Afamelanotide implants and narrow-band ultraviolet B phototherapy for the treatment of nonsegmental vitiligo in Asians. Journal of the American Academy of Dermatology. 2020 Jun 1;82(6):1517-9.
Lim HW, Grimes PE, Agbai O, Hamzavi I, Henderson M, Haddican M, Linkner RV, Lebwohl M. Afamelanotide and narrowband UV-B phototherapy for the treatment of vitiligo: a randomized multicenter trial. JAMA dermatology. 2015 Jan 1;151(1):42-50.
Communiqués
Vitiligo Communiqué IV
In this Vitiligo Communiqué IV, we expand on the long-term consequences, both physical and psychological, of vitiligo on patients, with a focus on phototypes IV–VI, and attempt to dispel some of the mischaracterisations surrounding vitiligo.
VIEW PDFVitiligo Communiqué III
Previously considered merely a cosmetic related condition, research now clearly demonstrates vitiligo is commonly associated with comorbidities including severe anxiety and depression. The third and final VITILIGO COMMUNIQUÉ, takes a look at the psychological impact of vitiligo on patients.
VIEW PDFVitiligo Communiqué II
In the second VITILIGO COMMUNIQUÉ, CLINUVEL looks closer at the role of narrowband UVB (NB-UVB) as the only standard of care at present in vitiligo, both the segmental and generalised form of the disease.
VIEW PDFVitiligo Communiqué I
What causes vitiligo? And what are the current treatments? VITILIGO COMMUNIQUÉ I reviews key concepts of a disease know to impact more than 1% of the global population.
VIEW PDF