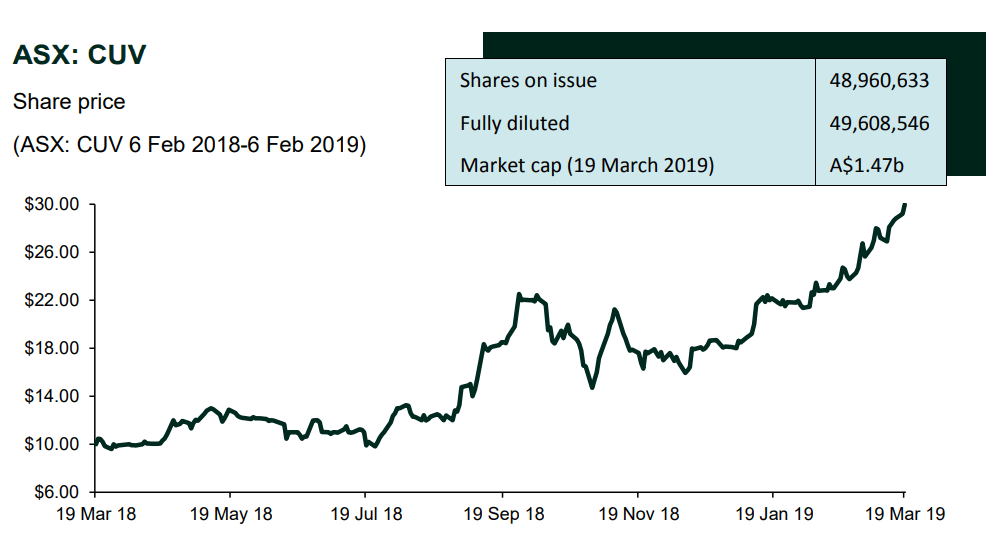
CLINUVEL Newsletter – March 2019
20 March 2019
Dear patients, shareholders, friends,
In the third Communiqué we are reviewing our EU operations, the impact of Brexit and looking at the year ahead.
FDA REVIEW
At the time of print CLINUVEL will find itself within four months of the US Prescription Drug User Fee Act (PDUFA) date, while our teams are making long nights and early mornings to address the US Food and Drug Agency’s (FDA’s) questions by given due dates. It is of interest that the turnaround of answers is often required within five to seven days. This requires our scientific teams in three continents to collaborate, coordinate and ensure that the answers are “formatted and published” by a third-party specialist service provider to meet the tight deadlines imposed. The process of answering often involves suppliers, manufacturers and service providers. Hours are limited and the agency’s demands require, at times, acrobatics from the CLINUVEL team. In order for all of us to perform, the regulatory, scientific, clinical, finance, communications, and media teams all work in concert and under pressure. We are all aware that this is a unique period of our professional lives and one that doesn’t come along often, hence the unified mindset to get this done.
I am utmost grateful to all the talented individuals we have within the Group working for one clinical cause. Professionally, for many of our managers this is a prime time in their professional lives and a unique opportunity to go through the US filing experience. While many final filings commonly require the sponsor to make a scientific case for benefit versus risk of the proposed technology, the FDA filing differs from European or other jurisdictions in that review processes, timelines and requirements differ. In February the FDA held its Mid-cycle Review Meeting, a moment in time when the various sections of the Division convene and review the dossier midway, as well as the additional information still required to arrive at a comprehensive benefit-risk assessment.

For the FDA – as said before – the times of reviewing a definitive filing (New Drug Application submission) is as exciting for them as it is for our staff. Certainly, in the Division of Dermatology and Dental Products it is not often that a new molecular entity (NME) is presented for marketing authorisation, and the staff of the Division go to great lengths to understand and analyse all parts of the drug product.
For our teams, this is a hectic time of having dinners at the office and finishing analyses and reviews until the wee hours of the morning, driven by one outcome and the awareness of the unique opportunity in their professional lives.
At CLINUVEL we all share the ambition of being able to offer juvenile EPP patients a treatment and normal life, and the only avenue to succeed in doing so is by obtaining approval for adult patients first. Unfortunately, and understandably so, drug development in adults precedes treatment in paediatric populations from a strict safety point of view. Therefore, the FDA outcome will determine our progress in the paediatric population of patients, and after more than a decade of supplying treatment to adults we gain confidence that a lower dose will be safe in children too
EUROPEAN DISTRIBUTION
As of 15 February, the first EPP patients in Europe have received SCENESSE® treatment for the calendar year.1 Our clinical and pharmacovigilance teams are now all too aware that patients also experience phototoxicity and anaphylactoid reactions during the winter months, and some physicians request drug treatment during the winter solstice.
The challenges this year are to manage the expansion of supply while maintaining the strict pharmacovigilance and reporting standards applicable to the EU pharmaceutical supply, all in a changing political climate. Solutions are being sought with National Competent Authorities to make SCENESSE® available through various avenues while CLINUVEL remains in full control of direct distribution to expert centres.
As we are entering the fourth season of clinical treatment after the marketing authorisation, it will be of interest to analyse how many EPP
patients continue treatment, emerging prescription patterns by expert physicians, and feedback from supporting medical staff.
In the meantime, we are expanding our team of clinical monitors and liaison managers in Europe, very much a function of growth of the Company. These staff facilitate the capture and monitoring of data under the marketing authorisation, ensure compliance with international guidelines and – in the case of liaison managers – the distribution of the product under controlled conditions directly to expert centres.
Imperative in continuous clinical treatment is that formal evaluations are being conducted independently, but also in collaboration between university centres and our scientific staff to understand the feedback and behavioural patterns of patients. We do this systematically and follow – in a ‘pseudonymised’ approach – each patient over time. Coupled with the feedback from porphyria patient associations a consistent picture and narrative is put together about the daily value of treatment provided.
Congruent with previous years, direct discussions with the healthcare professionals treating their patients are essential. This year we will learn first-hand in Berlin and Florence how experts experience the treatment and obtain direct feedback of their patients on longer-term treatment. This micro view is important to our research and development staff; at CLINUVEL we expose the laboratory and research staff to clinical experiences. We see it as important for all staff at each level to personally experience the collective goals we set and hear from the clinic first-hand how the lead drug affects patients and their families.
CLINUVEL GROWTH
In a controlled and gradual fashion, we are growing the Group, and recently we welcomed new scientists, administrative staff, and a new
investor relations manager to the Group. The integration of all new team members is critical to our long-term success, while the kernel of the Company takes on the task to pass on the modus operandi. Most apparent for this particular year is the corporate rhythm throughout the group of companies where all activities, events, developments and deadlines are coordinated and synchronised among all managers to make sure that we execute metronomically.
The team in Melbourne leads regulatory and financial oversight across the Group, with support for both functions expected to expand throughout the year.
CLINUVEL WORLDWIDE
CLINUVEL’s staff is stepping up its presence globally as demand is increasing to hear more details about the story. As the knowledge is spreading, US attention for SCENESSE® seems to be increasing, in view of the attention received at present, it is not surprising that our staff is asked to present the Company more often in public. At the same time patient organisations are inviting our clinical staff to brief on the treatment.
In the coming weeks, at the courtesy of four investment banks and one US publication house, CLINUVEL will present its story in front of institutional investors, analysts and a wider financial audience globally. The time prior to FDA outcome is opportune, since it provides us the ability to share our next phase for the US supply of SCENESSE®.
The schedule for April-July 2019 is:

These events not only provide the Company with exposure, but also valuable feedback as to where we sit relative to our peers. It is important for the team to benchmark our performance as well as learn from developments around us to be able to deliver on the mission we’ve set ourselves. Stakeholders who have attended recent Annual General Meetings of the Group will be familiar with management’s approach here.
BREXIT
It is little surprise that conversations worldwide at present are focused on events taking place in the United Kingdom. At time of writing – despite parliamentary resistance and votes casted against Theresa May’s Brexit proposal – the UK is still technically scheduled to depart from the European Union on 29 March, although the possibility of an extension is looming. In previous Communiqués we stressed the importance of this historic event not only politically, but economically for our sector. A number of changes are being implemented affecting drug supply within the European Union and single market.
Given the decision in 2015 to establish operations within the UK, there is a direct impact upon CLINUVEL, with functions now having been – or soon to be – moved out of England. Despite a diverse team in the UK – nearly half are EU nationals without British passports – none have decided to relocate as a result of Brexit and we will continue to maintain our UK office as an operational base for the foreseeable future. In the meantime, we are setting up other functions out of Ireland and mainland Europe.
In parallel, plans have been implemented to ensure continuity of supply to patients as our key focus, including seeking regulatory approvals to supply products cross-border under our controlled distribution program. This approach ensures for safety and product quality not to be affected while alterations to our processes are required while keeping the supply chain intact, all due to the unprecedented era in European history.
While we await further certainty from Whitehall, CLINUVEL will continue the migration of its operations to enable long-term compliance and, ultimately, patient treatment. This requires ongoing dialogue with the European Medicines Agency, National Competent Authorities and the European Commission and key suppliers to ensure a smooth transition. We also briefly discussed the newly introduced Falsified Medicines Directive EC62/2011 which affects the distribution of human medicines within the European Union.
Back to Brexit, depending on the terms of UK’s departure, the issue of continuing to be part of a single market or not will be essential to determine the value added tax on medicinal products, tariffs and conditions of importation. Our teams are watching closely as the economical divorce unfolds, while we prepare for various scenarios. It is feasible that a transition period will be implemented after following England’s exit according to Article 50.
NEXT PHASE
From evaluating key learnings from EU distribution, we are preparing similar quality management systems, pharmacovigilance, data collection, data storage reporting standards and other functions to be replicated for the US supply of SCENESSE®.
The challenges are abundant; however, much value is attributed to the key European managers in our team who are soon endowed with the responsibility of executing in the US. In many ways, the EU and Swiss distribution served our teams as a guided exercise while thinking ahead of how to efficiently manage future drug supply in the US, and all subject to FDA approval to market the pharmaceutical treatment.
As the PDUFA date approaches, our team has turned its attention to the challenges of a US roll-out and implementing learnings from the four years since EU marketing authorisation. We know our work is being watched closely by patients eagerly awaiting treatment, and will make more information public in due course.
FINANCIALS
On 26 February we posted CLINUVEL’s six monthly financial audited results – the half year ending 31 December 2018. The results have exceeded our expectations year on year while the price of the pharmaceutical product has not increased the past two years against rising supply and manufacturing costs. Although not obliged at this stage of financial reporting, the Company will continue to provide quarterly financial results in light of the seasonality of clinical demand and fluctuating cash receipts.
1 SCENESSE® (afamelanotide 16mg) is approved in Europe as an orphan medicinal product for the prevention of phototoxicity in adult patients with EPP. Information on the product can be found on CLINUVEL’s website at www.clinuvel.com