CLINUVEL Adds ACTH to Portfolio
| Melbourne, Australia, 08 November 2021 | ASX: XETRA-DAX: NASDAQ INTERNATIONAL DESIGNATION: |
CUV UR9 CLVLY |
Executive Summary
- ACTH – tradename NEURACTHEL® – added to CLINUVEL’s melanocortin portfolio
- Manufacturing agreement reached on scaled supply of cGMP grade ACTH
- Clinical opportunity: neurological, endocrinological, and degenerative disorders
- Commercial opportunity: global ACTH market est. >US$1.2B annually¹, CAGR 3.9%
- CLINUVEL translates its expertise in melanocortins and formulation development
CLINUVEL today announced that it has reached a longer term commercial agreement with one of its strategic partners for the scaled supply of adrenocorticotropic hormone (ACTH) drug substance to current Good Manufacturing Practices (cGMP) standard. Translating its expertise in melanocortins and formulation development, CLINUVEL will be commercialising NEURACTHEL® (ACTH) in multiple dosage forms for patients in need of medical treatment. The addition of NEURACTHEL® to CLINUVEL’s melanocortin portfolio forms part of the Company’s strategy to focus on a group of versatile hormone analogues.
ACTH belongs to the family of melanocortins, a group of bioactive hormones derived from the precursor peptide proopiomelanocortin (POMC), which is produced both in the pituitary gland and in peripheral tissues and skin. CLINUVEL’s lead pharmaceutical product afamelanotide is a synthetic analogue of another natural melanocortin in the human body, the α-melanocyte stimulating hormone (α-MSH), which is a derivative of ACTH. With the addition of NEURACTHEL®, CLINUVEL aims to take advantage of its expertise as a specialty pharmaceutical company focussed on the family of melanocortins.
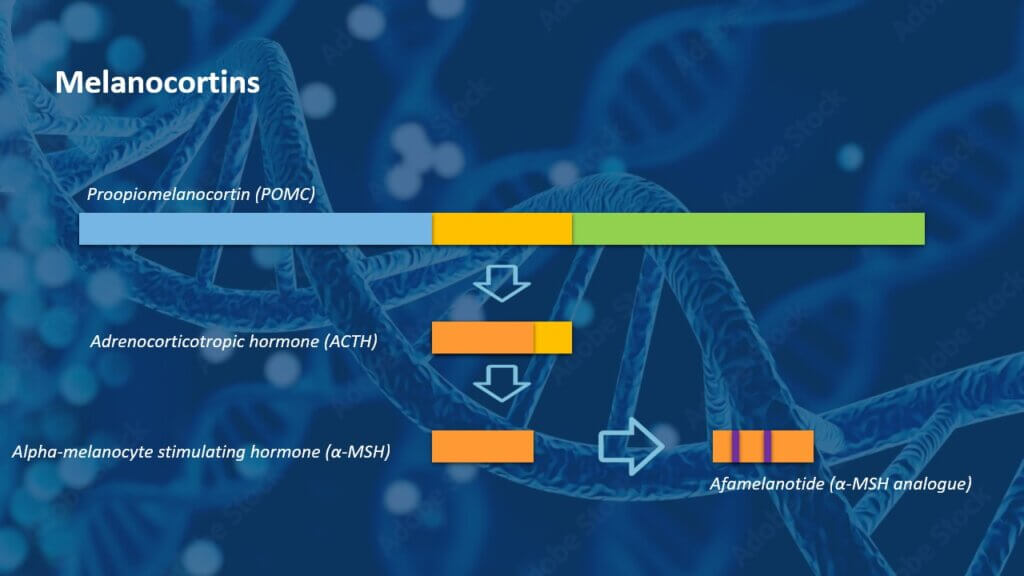
Figure 1 (left): Melanocortin hormones are derived from the precursor peptide proopiomelanocortin (POMC). ACTH and α-MSH are shorter, naturally occurring hormones. CLINUVEL has commercialised the novel drug afamelanotide, an analogue of α-MSH, as the controlled release injectable implant SCENESSE® which is approved by regulatory authorities in the USA, Europe and Australia.
Adrenocorticotropic Hormone (ACTH)
ACTH is a naturally occurring hormone which plays an important role in the production of cortisol, enabling the combat of stress and regulation of immune responses, maintenance of blood pressure, moderation of blood sugar, and regulation of metabolism.
Developed as a therapeutic agent in the 1950s, ACTH was first administered for human use as an animal derived hormone to influence the glucocorticoid secretion from the adrenal glands, and to treat a host of neurological and inflammatory diseases.
Pharmaceutical products containing ACTH or its analogues – including Cortrosyn®, Synacthen®, and Acthar® – are licensed in the USA, Europe and Asia-Pacific region in different dosage forms, including a solution with instant-release characteristics and a slow-release gel. ACTH analogues in liquid and gel formulations are used in severe chronic and acute neurological, endocrinological, and degenerative disorders such as Multiple Sclerosis.
Opportunity for Melanocortins to Address Unmet Clinical Need
CLINUVEL is pursuing its strategy to make melanocortins, including ACTH, available for patient populations with high unmet need (no effective or alternative treatment). The Company has long identified clinical potential for ACTH formulations beyond the currently approved clinical indications.
With additional patient populations growing unresponsive to corticosteroids and anti-inflammatory therapies, there is an increasing commercial opportunity to use NEURACTHEL®. In parallel, supply of ACTH in North America has been disrupted due to legal and financial challenges faced by one of the main manufacturers.
Worldwide, the ACTH market has been continuously growing, with a projected CAGR of 3.9% from 2021 to 2031. The global use of ACTH was valued at US$1.29 billion in 2020, and projected to reach US$1.91 billion by 2031.1 The driving factors for the expansion of the global ACTH market include the rise in the number of diseases to be diagnosed and treated by the bioactive hormone.
Clinuvel’s Clinical Focus on Melanocortins
CLINUVEL’s research and development led to the Company successfully commercialising the melanocortin analogue afamelanotide (SCENESSE® in erythropoietic protoporphyria; EPP). As part of afamelanotide’ s life-cycle management, various formulations are in advanced development stages, such as the new liquid presentations, PRÉNUMBRA® INSTANT and MODIFIED-RELEASE, for patients with acute and life-threatening conditions. Over 10,000 SCENESSE® doses have been administered to over 1,400 individuals.
In addition, the development of small melanocortin molecules will provide the Company with topical products (transdermal delivery), aiming to provide restorative and DNA reparative benefits.
Through CLINUVEL’s Research, Development and Innovation Centre in Singapore (VALLAURIX PTE LTD), a number of innovative controlled-release formulations are being advanced with targeted pharmacodynamic profiles. These formulations will be used as platforms for ACTH and other melanocortins.
Commentary
“The clinical and commercial opportunity to enter the ACTH market became more compelling as data came through, and was well timed with the maturity of our new formulation platforms,” CLINUVEL’s Vice President of Scientific Affairs, Dr Tim Zhao said. “We now pursue our vision to make NEURACTHEL® available to patient populations with high medical need in North America and Europe.
“Our expertise in peptide chemistry and polymers leads to today’s progress and strengthens the Company’s position as world leader in the development and clinical use of melanocortins. There is much work to be done, and given today’s commercial agreement, we are now ensuring availability of cGMP grade ACTH drug substance to be presented in our own formulations,” Dr Zhao said.
– END –
¹Adrenocorticotropic hormone (ACTH) market – global industry analysis, size, share, trends, and forecast, 2017-2031 by Transparency Market Research (TMR), 2021. Cortrosyn®, Synacthen® and Acthar® are trademarks of their respective owners.
About CLINUVEL PHARMACEUTICALS LIMITED
CLINUVEL PHARMACEUTICALS LTD (ASX: CUV; NASDAQ INTERNATIONAL DESIGNATION ADR: CLVLY; XETRA-DAX: UR9) is a global and diversified biopharmaceutical company focused on developing and commercialising treatments for patients with genetic, metabolic, and life-threatening disorders, as well as healthcare solutions for the general population. As pioneers in photomedicine and understanding the interaction of light and human biology, CLINUVEL’s research and development has led to innovative treatments for patient populations with a clinical need for systemic photoprotection, DNA repair and acute or life-threatening conditions. These patient groups range in size from 5,000 to 45 million worldwide. CLINUVEL’s lead compound, SCENESSE® (afamelanotide 16mg), was approved by the European Commission in 2014, the US Food and Drug Administration in 2019 and the Australian Therapeutic Goods Administration in 2020 for the prevention of phototoxicity (anaphylactoid reactions and burns) in adult patients with erythropoietic protoporphyria (EPP). More information on EPP can be found at http://www.epp.care. Headquartered in Melbourne, Australia, CLINUVEL has operations in Europe, Singapore and the USA. For more information please go to http://www.clinuvel.com.
SCENESSE®, PRÉNUMBRA® NEURACTHEL® are registered trademarks of CLINUVEL PHARMACEUTICALS LTD.
Authorised for ASX release by the Board of Directors of CLINUVEL PHARMACEUTICALS LTD
Head of Investor Relations
Mr Malcolm Bull, CLINUVEL PHARMACEUTICALS LTD
Investor Enquiries
https://www.clinuvel.com/investors/contact-us
Forward-Looking Statements
This release contains forward-looking statements, which reflect the current beliefs and expectations of CLINUVEL’s management. Statements may involve a number of known and unknown risks that could cause our future results, performance or achievements to differ significantly from those expressed or implied by such forward-looking statements. Important factors that could cause or contribute to such differences include risks relating to: our ability to develop and commercialise pharmaceutical products; the COVID-19 pandemic and/or other world, regional or national events affecting the supply chain for a protracted period of time, including our ability to develop, manufacture, market and sell biopharmaceutical products; competition for our products, especially SCENESSE® (afamelanotide 16mg), PRÉNUMBRA® or NEURACTHEL®; our ability to achieve expected safety and efficacy results in a timely manner through our innovative R&D efforts; the effectiveness of our patents and other protections for innovative products, particularly in view of national and regional variations in patent laws; our potential exposure to product liability claims to the extent not covered by insurance; increased government scrutiny in either Australia, the U.S., Europe, Israel, China and Japan of our agreements with third parties and suppliers; our exposure to currency fluctuations and restrictions as well as credit risks; the effects of reforms in healthcare regulation and pharmaceutical pricing and reimbursement; that the Company may incur unexpected delays in the outsourced manufacturing of SCENESSE®, PRÉNUMBRA® or NEURACTHEL® which may lead to it being unable to supply its commercial markets and/or clinical trial programs; any failures to comply with any government payment system (i.e. Medicare) reporting and payment obligations; uncertainties surrounding the legislative and regulatory pathways for the registration and approval of biotechnology and consumer based products; decisions by regulatory authorities regarding approval of our products as well as their decisions regarding label claims; our ability to retain or attract key personnel and managerial talent; the impact of broader change within the pharmaceutical industry and related industries; potential changes to tax liabilities or legislation; environmental risks; and other factors that have been discussed in our 2021 Annual Report. Forward-looking statements speak only as of the date on which they are made, and the Company undertakes no obligation, outside of those required under applicable laws or relevant listing rules of the Australian Securities Exchange, to update or revise any forward-looking statement, whether as a result of new information, future events or otherwise. More information on preliminary and uncertain forecasts and estimates is available on request, whereby it is stated that past performance is not an indicator of future performance.
www.clinuvel.com
Level 11
535 Bourke Street
Melbourne – Victoria, Australia, 3000
T +61 3 9660 4900
F +61 3 9660 4999