Appendix 4C & Activity Report
| Melbourne, Australia, 28 October 2021 | ASX: XETRA-DAX: NASDAQ INTERNATIONAL DESIGNATION: |
CUV UR9 CLVLY |
CLINUVEL PHARMACEUTICALS LTD today announced its Appendix 4C – Quarterly Cashflow Report and Activity Report for the period 01 July to 30 September 2021.
| Key Cash Flow Highlights Q1 FY20221 |
|
|---|---|
| Cash receipts | $17,785,000 |
| Cash expenditures | $6,297,000 |
| Net operating cash flow2 | +$11,560,000 |
| Cash reserves3 | +12.9% |
| Debt-free | |
|
|
Ongoing Clinical Treatment Delivers Positive Results
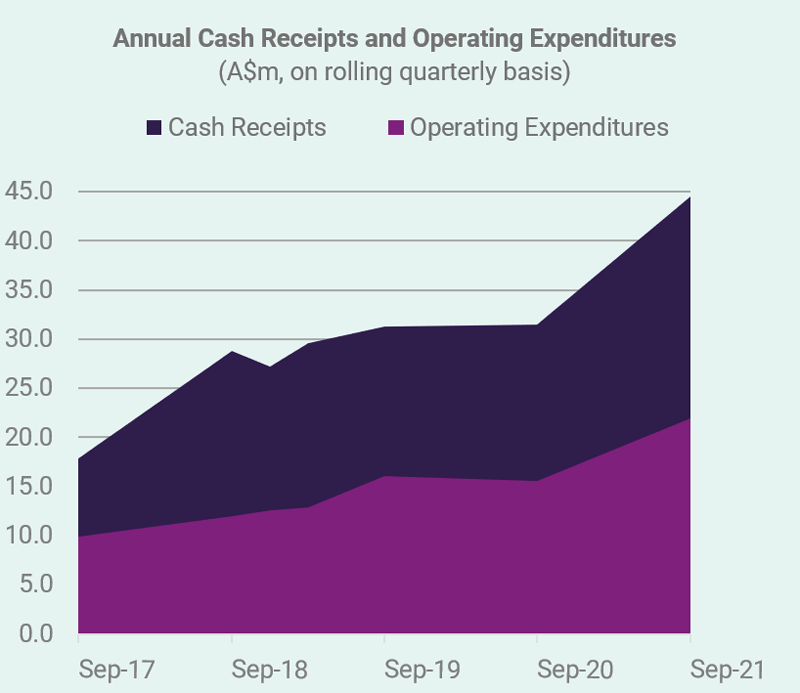
CLINUVEL has progressed its commercial operation, enabling greater access for adult patients with the rare metabolic disorder erythropoietic protoporphyria (EPP) to the Company’s drug SCENESSE® (afamelanotide 16mg).1 Operating in an environment where supply chains were disrupted and hospital services operated at limited capacity, resulting in restrictions and delays, the Company has collaborated with individual treatment centres in Europe, the USA and Israel to secure ongoing distribution and treatment, while continuous reimbursement of SCENESSE® delivered positive net operating cash flow for the quarter of $11,560,000.
For the September 2021 quarter, the Company’s cash receipts from commercial operations provide the latest indicator of the positive trend. The validation of the Company’s business model and long-term strategy is found in the fifth consecutive year of commercial operations which ended 30 June 2021. Consequent to a 43% increase in total revenues and a 123% increase to net profit before tax, the Board declared a fourth consecutive annual dividend during the quarter to express acknowledgement to shareholders supporting the Company’s financial management and overall direction.
CLINUVEL’s mission is to address untreated and neglected patient populations who have not yet received effective treatments, and to those groups in society who would benefit from targeted personal care products. The Company’s strategy focusses on developing pharmaceutical treatments and non-prescription healthcare solutions. The non-prescriptive healthcare solution line aims to reach groups at highest risk of solar damage and skin cancer due to exposure to ultraviolet and high energy visible light. The near-term aim of the strategy is to see the Company establish a sustainable and diversified healthcare business.
Increasing Cash Receipts and Net Cash
CLINUVEL has reported cash receipts of $17,785,000 for the September quarter of 2021. This is the Company’s highest recorded cash receipt result for a quarter. This increase also reflects the initial growth in the demand for SCENESSE® in the United States and its acceptance by US insurers to reimburse the drug under Prior Authorization arrangements, and as part of the inclusion on formulary lists. Cash inflows from European expert centres also corresponded with strong treatment demand in the months of more intense light exposure.
Net operating cash flows were $11,560,000 for the quarter. Total cash reserves were impacted by CLINUVEL paying, on 17 September 2021, a full year unfranked dividend distribution of $0.025 per share, translating to a total $1,235,000 distribution payment to registered holders.
Financial Commentary on Results
“It is now 18 months since the first US order was placed and, parallel to first-time patients receiving the drug, we are also seeing patients return on a consistent basis to seek continuous, year-round treatment,” CLINUVEL’s Chief Financial Officer, Mr Darren Keamy said. “Insurers are accepting of the medical necessity for the recurring treatment of SCENESSE® and are providing the requisite financial support for their EPP patient constituents.
“The quarterly cash flow result reflects the growth in our commercial program. Not only is this an exciting position for the Company, it also requires longer term planning of our use of assets while maintaining a disciplined approach to cost management.
“In planning for the further expansion of the Company on multiple fronts, we are investing in talented staff, pipeline development and operational infrastructure. We will continue to manage risk as we continue along this exciting growth trajectory,” Mr Keamy said.
Expenditures and Reinvestments
Expenditures from operating activities were $6,297,000 for the September quarter. These were characterised by payments towards manufacturing, supply costs of raw materials and finished goods. Amid increasing cost pressures across the entire supply chain, the Company is undertaking a program to fortify stock levels to meet expected future patient demand and to support the expanded range of R&D initiatives. Investment in the Company’s talent pool is reflected in the staffing costs. Headcount is expected to continue to increase in this financial year in support of the Company’s expansion.
Review of Key Activities This Quarter
In addition to increased commercial distribution of SCENESSE® for EPP patients in the September quarter, the Company worked to progress the expanding clinical program. Specifically, on key programs:
DNA Repair Program
- The focus was on preparations to commence the first global studies in the rare genetic disorder xeroderma pigmentosum (XP), a life-threatening disorder due to the patients’ inability to repair UV-provoked DNA skin damage. This work culminated in the announcement early in the current quarter that all necessary regulatory and ethics committee approvals to commence a new study had been granted. Last week, as the local hospitals resumed operations under full capacity, the Company announced the treatment of the first XP patient (CUV156 study). The patient is diagnosed with the XP-C complementation.
Arterial Ischaemic Stroke (AIS)
- The Company reported that afamelanotide was well tolerated by the first stroke patients in the CUV801 study. Three AIS patients had been treated. No patient experienced drug-related adverse effects and all were discharged from critical care. COVID-19 related lockdowns during the quarter delayed further progress at the treating hospital in Australia, but clinicians are now proceeding to treat the next three of the planned six patients of the study.
Vitiligo
- The Company continued to prepare and coordinate preparatory work to reach agreement with the US Food and Drug Administration on the design of a new North American vitiligo study. The focus is on patients of darker skin colour, for whom the need for repigmentation treatment is regarded as the greatest due to the loss of identity. CLINUVEL expects to provide a further update on this program in the coming months.
Following the 26 August 2021 announcement of the record revenues and profit achieved in the financial year ended 30 June 2021, the Company responded to requests for media interviews and commentary captured in a communications program, which included institutional investor teleconferences and an Annual Results Webinar.
All of the Company’s announcements and key media interviews in the September quarter 2021 are available on the CLINUVEL website, with other updates available on the CLINUVEL News website.
Although the Company is no longer obligated under ASX Listing Rules to publish quarterly cash flow results, it elects to continue to do so to keep its global investors updated regularly. A copy of the Appendix 4C – Quarterly Cash Flow Report for the second quarter of FY2021 is attached.
Pursuant to Listing Rule 4.7C and as disclosed in Item 6.1 to the attached Appendix 4C, $741,000 (inclusive of non-monetary benefits) were recorded in respect to Non-Executive Directors’ fees, Managing Director’s fees and non-monetary benefits.
– End –
SCENESSE® (afamelanotide 16mg) is approved in the European Union and Australia as an orphan medicinal product for the prevention of phototoxicity in adult patients with erythropoietic protoporphyria (EPP). SCENESSE® is approved in the USA to increase “pain-free” light exposure in adult EPP patients with a history of phototoxicity. Information on the product can be found on CLINUVEL’s website at www.clinuvel.com.
About CLINUVEL PHARMACEUTICALS LIMITED
CLINUVEL PHARMACEUTICALS LTD (ASX: CUV; NASDAQ INTERNATIONAL DESIGNATION ADR: CLVLY; XETRA-DAX: UR9) is a global and diversified biopharmaceutical company focused on developing and commercialising treatments for patients with genetic, metabolic, and life-threatening disorders, as well as healthcare solutions for the general population. As pioneers in photomedicine and understanding the interaction of light and human biology, CLINUVEL’s research and development has led to innovative treatments for patient populations with a clinical need for systemic photoprotection, DNA repair and acute or life-threatening conditions. These patient groups range in size from 5,000 to 45 million worldwide. CLINUVEL’s lead compound, SCENESSE® (afamelanotide 16mg), was approved by the European Commission in 2014, the US Food and Drug Administration in 2019 and the Australian Therapeutic Goods Administration in 2020 for the prevention of phototoxicity (anaphylactoid reactions and burns) in adult patients with erythropoietic protoporphyria (EPP). More information on EPP can be found at http://www.epp.care. Headquartered in Melbourne, Australia, CLINUVEL has operations in Europe, Singapore and the USA. For more information please go to http://www.clinuvel.com.
SCENESSE® and PRÉNUMBRA® are registered trademarks of CLINUVEL PHARMACEUTICALS LTD.
Authorised for ASX release by the Board of Directors of CLINUVEL PHARMACEUTICALS LTD
Media Enquiries
Monsoon Communications
Mr Rudi Michelson, 61 411 402 737, rudim@monsoon.com.au
Head of Investor Relations
Mr Malcolm Bull, CLINUVEL PHARMACEUTICALS LTD
Investor Enquiries
https://www.clinuvel.com/investors/contact-us
Forward-Looking Statements
This release contains forward-looking statements, which reflect the current beliefs and expectations of CLINUVEL’s management. Statements may involve a number of known and unknown risks that could cause our future results, performance, or achievements to differ significantly from those expressed or implied by such forward-looking statements. Important factors that could cause or contribute to such differences include risks relating to: our ability to develop and commercialise pharmaceutical products, the COVID-19 pandemic affecting the supply chain for a protracted period of time, including our ability to develop, manufacture, market and sell biopharmaceutical products; competition for our products, especially SCENESSE® (afamelanotide 16mg); our ability to achieve expected safety and efficacy results through our innovative R&D efforts; the effectiveness of our patents and other protections for innovative products, particularly in view of national and regional variations in patent laws; our potential exposure to product liability claims to the extent not covered by insurance; increased government scrutiny in either Australia, the U.S., Europe, China and Japan of our agreements with third parties and suppliers; our exposure to currency fluctuations and restrictions as well as credit risks; the effects of reforms in healthcare regulation and pharmaceutical pricing and reimbursement; that the Company may incur unexpected delays in the outsourced manufacturing of SCENESSE® which may lead to it being unable to supply its commercial markets and/or clinical trial programs; any failures to comply with any government payment system (i.e. Medicare) reporting and payment obligations; uncertainties surrounding the legislative and regulatory pathways for the registration and approval of biotechnology based products; decisions by regulatory authorities regarding approval of our products as well as their decisions regarding label claims; any failure to retain or attract key personnel and managerial talent; the impact of broader change within the pharmaceutical industry and related industries; potential changes to tax liabilities or legislation; environmental risks; and other factors that have been discussed in our 2020 Annual Report. Forward-looking statements speak only as of the date on which they are made, and the Company undertakes no obligation, outside of those required under applicable laws or relevant listing rules of the Australian Securities Exchange, to update or revise any forward-looking statement, whether as a result of new information, future events or otherwise. More information on the forecasts and estimates is available on request. Past performance is not an indicator of future performance.
www.clinuvel.com
Level 11
535 Bourke Street
Melbourne – Victoria, Australia, 3000
T +61 3 9660 4900
F +61 3 9660 4999
Appendix 4C
Quarterly cash flow report for entities subject to Listing Rule 4.7B
Name of entity
CLINUVEL PHARMACEUTICALS LIMITED
ABN
88 089 644 119
Quarter ended (“current quarter”)
30 SEPTEMBER 2021
| Consolidated statement of cash flows | Current quarter $A’000 |
Year to date (3 months) $A’000 |
|
|---|---|---|---|
| 1 | Cash flows from operating activities | ||
| 1.1 | Receipts from customers | 17,785 | 17,785 |
| 1.2 | Payments for | ||
| (a) research and development | (206) | (206) | |
| (b) product manufacturing and operating costs | (2,673) | (2,673) | |
| (c) advertising and marketing | (69) | (69) | |
| (d) leased assets | (73) | (73) | |
| (e) staff costs | (2,573) | (2,573) | |
| (f) administration and corporate costs | (737) | (737) | |
| 1.3 | Dividends received (see note 3) | – | – |
| 1.4 | Interest received | 63 | 63 |
| 1.5 | Interest and other costs of finance paid | (11) | (11) |
| 1.6 | Income taxes paid | – | – |
| 1.7 | Government grants and tax incentives | 10 | 10 |
| 1.8 | Other (provide details if material) | 44 | 44 |
| 1.9 | Net cash from / (used in) operating activities | 11,560 | 11,560 |
| 2 | Cash flows from investing activities | ||
| 2.1 | Payments to acquire or for: | ||
| (a) entities | |||
| (b) businesses | – | – | |
| (c) property, plant and equipment | (21) | (21) | |
| (d) investments | – | – | |
| (e) intellectual property | – | – | |
| (f) other non-current assets | – | – | |
| 2.2 | Proceeds from disposal of: | – | – |
| (a) entities | |||
| (b) businesses | – | – | |
| (c) property, plant and equipment | – | – | |
| (d) investments | – | – | |
| (e) intellectual property | – | – | |
| (f) other non-current assets | – | – | |
| 2.3 | Cash flows from loans to other entities | – | – |
| 2.4 | Dividends received (see note 3) | – | – |
| 2.5 | Other (provide details if material) | – | – |
| 2.6 | Net cash from / (used in) investing activities | (21) | (21) |
| 3 | Cash flows from financing activities | ||
| 3.1 |
Proceeds from issues of equity securities (excluding convertible debt securities) |
– | – |
| 3.2 | Proceeds from issue of convertible debt securities | – | – |
| 3.3 | Proceeds from exercise of options | – | – |
| 3.4 |
Transaction costs related to issues of equity securities or convertible debt securities |
– | – |
| 3.5 | Proceeds from borrowings | – | – |
| 3.6 | Repayment of borrowings | (62) | (62) |
| 3.7 | Transaction costs related to loans and borrowings | – | – |
| 3.8 | Dividends paid | (1,235) | (1,235) |
| 3.9 | Other (provide details if material) | – | – |
| 3.10 | Net cash from / (used in) financing activities | (1,297) | (1,297) |
| 4 |
Net increase / (decrease) in cash and cash equivalents for the period |
||
| 4.1 | Cash and cash equivalents at beginning of period | 82,691 | 82,691 |
| 4.2 | Net cash from / (used in) operating activities (item 1.9 above) | 11,560 | 11,560 |
| 4.3 | Net cash from / (used in) investing activities (item 2.6 above) | (21) | (21) |
| 4.4 | Net cash from / (used in) financing activities (item 3.10 above) | (1,297) | (1,297) |
| 4.5 | Effect of movement in exchange rates on cash held | 450 | 450 |
| 4.6 | Cash and cash equivalents at end of period | 93,383 | 93,383 |
| 5 | Reconciliation of cash and cash equivalents at the end of the quarter (as shown in the consolidated statement of cash flows) to the related items in the accounts |
Current quarter $A’000 |
Previous quarter $A’000 |
|---|---|---|---|
| 5.1 | Bank balances | 39,307 | 39,307 |
| 5.2 | Call deposits | 53,800 | 53,800 |
| 5.3 | Bank overdrafts | – | – |
| 5.4 | Other (provide details) | 276 | 276 |
| 5.5 | Cash and cash equivalents at end of quarter (should equal item 4.6 above) | 93,383 | 93,383 |
| 6 | Payments to related parties of the entity and their associates | Current quarter $A’000 |
|---|---|---|
| 6.1 | Aggregate amount of payments to related parties and their associates included in item 1 | 741 |
| 6.2 | Aggregate amount of payments to related parties and their associates included in item 2 | |
| Note: if any amounts are shown in items 6.1 or 6.2, your quarterly activity report must include a description of, and an explanation for, such payments. | ||
| 7 | Financing facilities
Note: the term “facility” includes all forms of financing arrangements available to the entity. Add notes as necessary for an understanding of the sources of finance available to the entity. |
Total facility amount at quarter end $A’000 |
Amount drawn at quarter end $A’000 |
|---|---|---|---|
| 7.1 | Loan facilities | ||
| 7.2 | Credit standby arrangements | ||
| 7.3 | Other (please specify) | ||
| 7.4 | Total financing facilities | ||
| 7.5 | Unused financing facilities available at quarter end | ||
| 7.6 | Include in the box below a description of each facility above, including the lender, interest rate, maturity date and whether it is secured or unsecured. If any additional financing facilities have been entered into or are proposed to be entered into after quarter end, include a note providing details of those facilities as well. | ||
| 8 | Estimated cash available for future operating activities | $A’000 |
|---|---|---|
| 8.1 | Net cash from / (used in) operating activities (item 1.9) | 11,560 |
| 8.2 | Cash and cash equivalents at quarter end (item 4.6) | 93,383 |
| 8.3 | Unused finance facilities available at quarter end (item 7.5) | 0 |
| 8.4 | Total available funding (item 8.2 + item 8.3) | 93,383 |
| 8.5 | Estimated quarters of funding available (item 8.4 divided by item 8.1) | N/A |
| Note: if the entity has reported positive net operating cash flows in item 1.9, answer item 8.5 as “N/A”. Otherwise, a figure for the estimated quarters of funding available must be included in item 8.5. | ||
| 8.6 | If item 8.5 is less than 2 quarters, please provide answers to the following questions: | |
| 8.6.1 | Does the entity expect that it will continue to have the current level of net operating cash flows for the time being and, if not, why not? | |
| Answer: N/A | ||
| 8.6.2 | Has the entity taken any steps, or does it propose to take any steps, to raise further cash to fund its operations and, if so, what are those steps and how likely does it believe that they will be successful? | |
| Answer: N/A | ||
| 8.6.3 | Does the entity expect to be able to continue its operations and to meet its business objectives and, if so, on what basis? | |
| Answer: N/A | ||
| Note: where item 8.5 is less than 2 quarters, all of questions 8.6.1, 8.6.2 and 8.6.3 above must be answered. | ||
Compliance statement
- This statement has been prepared in accordance with accounting standards and policies which comply with Listing Rule 19.11A.
- This statement gives a true and fair view of the matters disclosed.
Date: 28 October 2021
Authorised by: MR DARREN KEAMY
(Name of body or officer authorising release – see note 4)
Notes
- This quarterly cash flow report and the accompanying activity report provide a basis for informing the market about the entity’s activities for the past quarter, how they have been financed and the effect this has had on its cash position. An entity that wishes to disclose additional information over and above the minimum required under the Listing Rules is encouraged to do so.
- If this quarterly cash flow report has been prepared in accordance with Australian Accounting Standards, the definitions in, and provisions of, AASB 107: Statement of Cash Flows apply to this report. If this quarterly cash flow report has been prepared in accordance with other accounting standards agreed by ASX pursuant to Listing Rule 19.11A, the corresponding equivalent standard applies to this report.
- Dividends received may be classified either as cash flows from operating activities or cash flows from investing activities, depending on the accounting policy of the entity.
- If this report has been authorised for release to the market by your board of directors, you can insert here: “By the board”. If it has been authorised for release to the market by a committee of your board of directors, you can insert here: “By the [name of board committee – eg Audit and Risk Committee]”. If it has been authorised for release to the market by a disclosure committee, you can insert here: “By the Disclosure Committee”.
- If this report has been authorised for release to the market by your board of directors and you wish to hold yourself out as complying with recommendation 4.2 of the ASX Corporate Governance Council’s Corporate Governance Principles and Recommendations, the board should have received a declaration from its CEO and CFO that, in their opinion, the financial records of the entity have been properly maintained, that this report complies with the appropriate accounting standards and gives a true and fair view of the cash flows of the entity, and that their opinion has been formed on the basis of a sound system of risk management and internal control which is operating effectively.