Appendix 4C & Activity Report
| Melbourne, Australia, 31 October 2022 | ASX: XETRA-DAX: ADR Level 1: |
CUV UR9 CLVLY |
The Appendix 4C lodged earlier today reported in item 1.9 a “Net cash from / (used in) operating activities” result of 117,338 for the quarter and year to date. This number should read 17,338.
This has now been corrected and reflected in the Appendix 4C attached.
CLINUVEL PHARMACEUTICALS LTD released its Appendix 4C – Quarterly Cashflow
Report and Activity Report for the period 01 July to 30 September 2022.
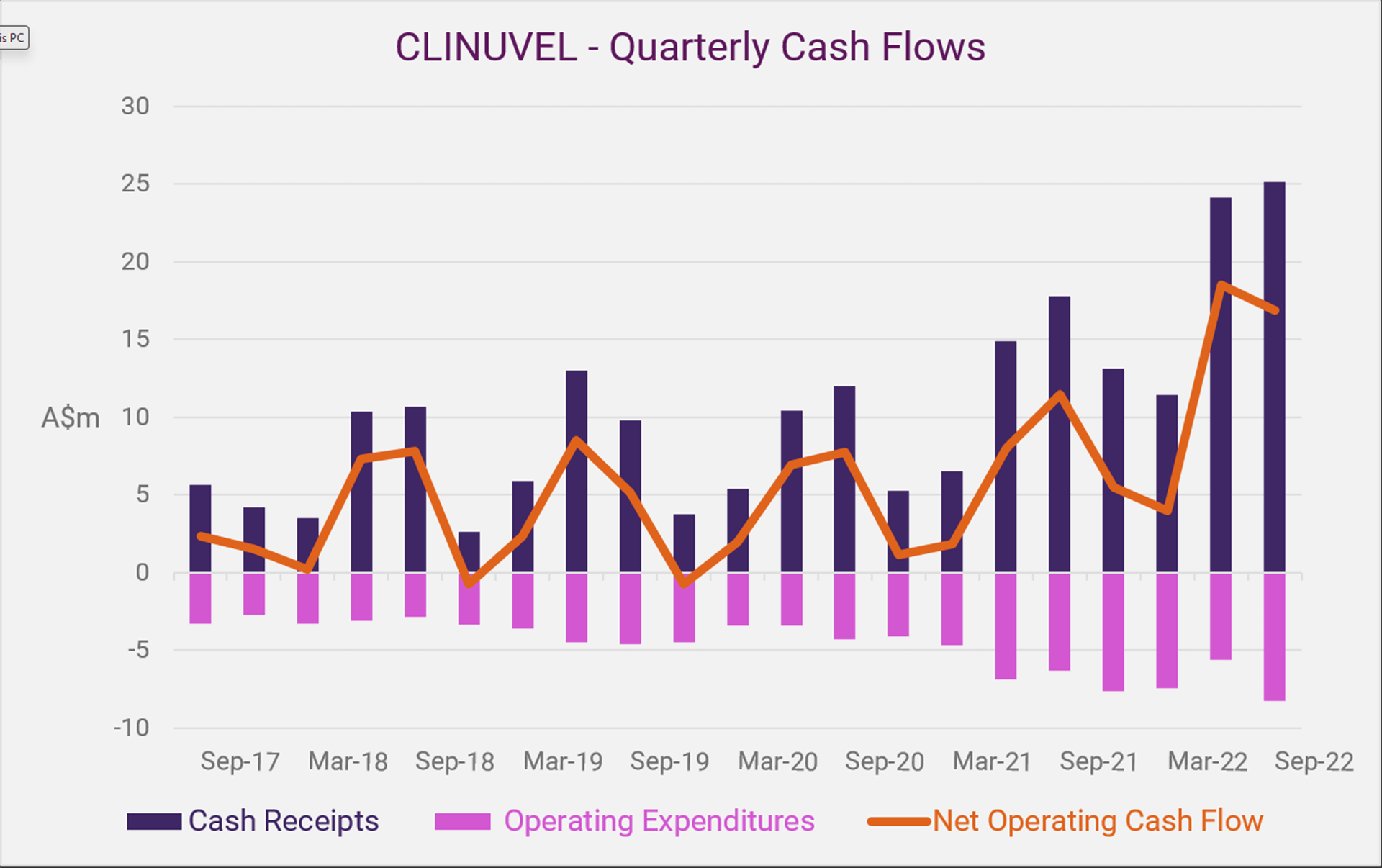
| Highlights Cash Flow | ||
|---|---|---|
| Q1 FY20231 | ||
| Cash receipts2 | $25,512,000 | |
| Operating Cash expenditures | $8,270,000 | |
| Net operating cash flow2 | +$17,338,000 | |
| Cash reserves3 | +13.3% | |
|
||
RISING CUSTOMER RECEIPTS, CONTROLLED EXPENDITURES
CLINUVEL recorded its highest quarterly results from customer receipts since the commencement of commercial operations in Europe in June 2016 and United States in April 2020. New patient uptake of CLINUVEL’s drug SCENESSE® (afamelanotide 16mg) coincided with peak treatment demand from both continents. CLINUVEL’s receipts from customers for the July to September quarter were $25.152 million, with net operating expenditures of $8.270 million and net operating cashflow totalling $17.338 million.
The quarter-on-quarter increase of overall net operating expenditures (47%) reflects the impact of the timing of certain payments as well as a controlled increased in overall expenditures as CLINUVEL expands its R&D activities. On an annual basis, operating expenditures demonstrates a 31.9% increase for the 12 months to 30 September 2022, indicating an ongoing commitment to re-invest in the business.
FULL YEAR DIVIDEND
Total cash reserves were impacted by CLINUVEL paying, on 21 September 2022, a
full year unfranked dividend distribution of $0.04 per share, translating to a $1,976,000 payment to registered holders. This was a 60% increase to the dividend distribution made in September 2021. The strong performance of the Company for the 2021/22 financial year allowed the Board to declare the increase in dividend, most of all recognising the loyalty and patience of long-term shareholders.
“The positive net cash inflow results reflects the demand over the spring and summer months in the northern hemisphere, the efficiency of the Company’s commercial infrastructure, coupled with a focus on containing cost increases,” CLINUVEL’s Chief Financial Officer, Darren Keamy said.
“The growth in the cash position enables management to dedicate itself to pursuing further strategic initiatives, while research and development are ongoing to expand the Company’s pipeline. Global economic conditions are dire and an energy crisis is looming in many parts of the world, yet we are on course to realise CLINUVEL’s growth plans, allowing us to come out stronger when the global economies recover.”
POSITION TO SELF-FINANCE GROWTH
CLINUVEL has communicated a strategic commitment to spend $175m on the growth of the business over the five years to 30 June 2025. Expenses are steadily increasing but well within the window projected for its Pharmaceutical and Healthcare Solutions Divisions to expand clinical use of SCENESSE® and to add new products. The Company’s investments remain on track with its forecast with just over $55 million spent in FY2021/22, and therefore, around $120 million to be expended over the next three years. This excludes capital expenditures.
KEY ACTIVITIES
Key activities in the September quarter 2022 are summarised below.
Commercial Operations
During the past quarter, treatment was extended to more patients and the close
relationships with healthcare professionals, insurers, and government agencies
continued to facilitate access to treatment for erythropoietic protoporphyria
(EPP) patients.
Progress of Clinical Programs
CLINUVEL is focused on an expanded range of targeted clinical development
programs evaluating the safety and efficacy of melanocortins – including
afamelanotide – for patients with genetic, metabolic, life-threatening and
acute disorders. A summary of the clinical programs is provided below:
| Study | Status |
|---|---|
| CUV156 adult xeroderma pigmentosum C (XP-C) patients | Study ongoing. Results expected in 2022. |
| CUV151 disease free adult subjects |
First subject dosed in February 2022. Results expected in 2022. |
|
CUV152 adult xeroderma pigmentosum V & C (XP-V/XP-C) patients |
First XP-V patient dosed in March. Results expected in 2022. |
| Study | Status | ||
|---|---|---|---|
|
Study complete, results announced. Afamelanotide was evaluated as safe in mild to moderate AIS. Neurological function, as measured by the National Institutes of Health Stroke Scale (NIHSS) improved in five patients, brain scans (MRI-FLAIR) in all six patients showed reduction of affected hypoxic tissue, and there was strong functional recovery in all five surviving patients. |
In July 2022, the Company announced that PRÉNUMBRA® Instant, a fast-acting, flexible afamelanotide would be used in the next stroke study, CUV803, expected to commence in 2022. |
| Study | Status |
|---|---|
|
CUV104 adult vitiligo patients with darker skin complexion (Fitzpatrick IV-VI) |
Study design agreed with the US Food and Drug Administration (FDA). Institutional Review Board approved CUV104 study to focus on patients of darker skin complexion (Fitzpatrick types IV-VI). Afamelanotide to be assessed as a monotherapy for systemic repigmentation of vitiligo patients. In October 2022, the Company announced that the |
Healthcare Solutions
CLINUVEL differentiates itself from most pharmaceutical companies by translating its expertise and technology (the class of melanocortins) to non-prescription products for wider use and for specialised populations in need of strong and long term skin care. These populations are characterised as being at Highest Risk of photodamage and skin cancers.
CLINUVEL continues to work towards the launch of a range of dermatocosmetic products aimed at specialised populations at highest risk from exposure to ultraviolet (UV) and high energy visible (HEV) light. The first product CYACÊLLE® provides polychromatic protection against light in the invisible and visible spectrum. Research & development of future product lines is underway. These products will assist in DNA repair of affected skin, while aiming to stabilise melanogenesis.
Ahead of formal product launch, staggered digital marketing campaigns are underway to establish specialized audiences at highest risk of solar damage and prone to skin cancers and melanoma. New digital platforms, including LightSkinScience.com, and dedicated social media channels have been launched, with a team of CLINUVEL Ambassadors (CUVAs) engaged to educate and build connectivity with the targeted audiences.
Other Activities and Announcements
The Company’s Annual Report for the past financial year (ended 30 June 2022) was
announced on 30 August 2022 and, following a briefing to shareholders first in Basel and later in Monaco, the Company released its fifth Strategic Update on 19 September. The Company’s announcements in the September quarter 2022 are available on the CLINUVEL website, with other updates available on the CLINUVEL News website.
Although the Company is no longer obligated under ASX Listing Rules to publish quarterly cash flow results, it elects to continue to do so to keep its global investors updated regularly. A copy of the Appendix 4C – Quarterly Cash Flow Report for the final quarter of FY2022 is attached.
Pursuant to Listing Rule 4.7C and as disclosed in Item 6.1 to the attached
Appendix 4C, $2,648,000 were recorded in respect to Non-Executive Directors’
fees, Managing Director’s fees and payments towards non-monetary benefits
(inclusive of short-term incentive and long-term retention benefit entitlement
to Managing Director).
– End –
1 SCENESSE® (afamelanotide 16mg) is approved in the European Union and Australia as an orphan medicinal product for the prevention of phototoxicity in adult patients with erythropoietic protoporphyria (EPP). SCENESSE® is approved in the USA to increase “pain-free” light exposure in adult EPP patients with a history of phototoxicity. Information on the product can be found on CLINUVEL’s website at www.clinuvel.com.
About CLINUVEL PHARMACEUTICALS LIMITED
CLINUVEL (ASX: CUV; ADR LEVEL 1: CLVLY; XETRA-DAX: UR9) is a global specialty pharmaceutical group focused on developing and commercialising treatments for patients with genetic, metabolic, systemic, and life-threatening, acute disorders, as well as healthcare solutions for the general population. As pioneers in photomedicine and the family of melanocortin peptides, CLINUVEL’s research and development has led to innovative treatments for patient populations with a clinical need for systemic photoprotection, DNA repair, repigmentation and acute or life-threatening conditions who lack alternatives.
CLINUVEL’s lead therapy, SCENESSE® (afamelanotide 16mg), is approved for commercial distribution in Europe, the USA, Israel and Australia as the world’s first systemic photoprotective drug for the prevention of phototoxicity (anaphylactoid reactions and burns) in adult patients with erythropoietic protoporphyria (EPP). Headquartered in Melbourne, Australia, CLINUVEL has operations in Europe, Singapore and the USA. For more information, please go to https://www.clinuvel.com.
SCENESSE®, PRÉNUMBRA® and NEURACTHEL® are registered trademarks of CLINUVEL PHARMACEUTICALS LTD.
Authorised for ASX release by the Board of Directors of CLINUVEL PHARMACEUTICALS LTD
Media Enquiries
Monsoon Communications
Mr Rudi Michelson, 61 411 402 737, rudim@monsoon.com.au
Head of Investor Relations
Mr Malcolm Bull, CLINUVEL PHARMACEUTICALS LTD
Investor Enquiries
https://www.clinuvel.com/investors/contact-us
Forward-Looking Statements
This release contains forward-looking statements, which reflect the current beliefs and expectations of CLINUVEL’s management. Statements may involve a number of known and unknown risks that could cause our future results, performance or achievements to differ significantly from those expressed or implied by such forward-looking statements. Important factors that could cause or contribute to such differences include risks relating to: our ability to develop and commercialise pharmaceutical products; the COVID-19 pandemic and/or other world, regional or national events affecting the supply chain for a protracted period of time, including our ability to develop, manufacture, market and sell biopharmaceutical products; competition for our products, especially SCENESSE® (afamelanotide 16mg), PRÉNUMBRA® or NEURACTHEL®; our ability to achieve expected safety and efficacy results in a timely manner through our innovative R&D efforts; the effectiveness of our patents and other protections for innovative products, particularly in view of national and regional variations in patent laws; our potential exposure to product liability claims to the extent not covered by insurance; increased government scrutiny in either Australia, the U.S., Europe, Israel, China and Japan of our agreements with third parties and suppliers; our exposure to currency fluctuations and restrictions as well as credit risks; the effects of reforms in healthcare regulation and pharmaceutical pricing and reimbursement; that the Company may incur unexpected delays in the outsourced manufacturing of SCENESSE®, PRÉNUMBRA® or NEURACTHEL® which may lead to it being unable to supply its commercial markets and/or clinical trial programs; any failures to comply with any government payment system (i.e. Medicare) reporting and payment obligations; uncertainties surrounding the legislative and regulatory pathways for the registration and approval of biotechnology and consumer based products; decisions by regulatory authorities regarding approval of our products as well as their decisions regarding label claims; our ability to retain or attract key personnel and managerial talent; the impact of broader change within the pharmaceutical industry and related industries; potential changes to tax liabilities or legislation; environmental risks; and other factors that have been discussed in our 2021 Annual Report. Forward-looking statements speak only as of the date on which they are made, and the Company undertakes no obligation, outside of those required under applicable laws or relevant listing rules of the Australian Securities Exchange, to update or revise any forward-looking statement, whether as a result of new information, future events or otherwise. More information on preliminary and uncertain forecasts and estimates is available on request, whereby it is stated that past performance is not an indicator of future performance.
www.clinuvel.com
Level 11
535 Bourke Street
Melbourne – Victoria, Australia, 3000
T +61 3 9660 4900
F +61 3 9660 4999
Appendix 4C
Quarterly cash flow report for entities subject to Listing Rule 4.7B
Name of entity
CLINUVEL PHARMACEUTICALS LIMITED
ABN
88 089 644 119
Quarter ended (“current quarter”)
31 SEPTEMBER 2022
| Consolidated statement of cash flows | Current quarter $A’000 |
Year to date (3 months) $A’000 |
|
|---|---|---|---|
| 1 | Cash flows from operating activities | ||
| 1.1 | Receipts from customers | 25,512 | 25,512 |
| 1.2 | Payments for | ||
| research and development | (532) | (532) | |
| product manufacturing and operating costs | (1,235) | (1,235) | |
| advertising and marketing | (229) | (229) | |
| leased assets | (77) | (77) | |
| staff costs | (5,107) | (5,107) | |
| administration and corporate costs | (1,096) | (3,096) | |
| 1.3 | Dividends received (see note 3) | – | – |
| 1.4 | Interest received | 88 | 88 |
| 1.5 | Interest and other costs of finance paid | (12) | (12) |
| 1.6 | Income taxes paid | (25) | (25) |
| 1.7 | Government grants and tax incentives | 0 | 210 |
| 1.8 | Other (provide details if material) | 42 | 42 |
| 1.9 | Net cash from / (used in) operating activities | 17,338 | 17,338 |
| 2 | Cash flows from investing activities | ||
| 2.1 | Payments to acquire or for: | ||
| (a) entities | – | – | |
| businesses | – | – | |
| property, plant and equipment | (117) | (117) | |
| investments | – | – | |
| intellectual property | – | – | |
| other non-current assets | – | – | |
| 2.2 | Proceeds from disposal of: | – | – |
| (b) entities | |||
| businesses | – | – | |
| property, plant and equipment | – | – | |
| investments | – | – | |
| intellectual property | – | – | |
| other non-current assets | – | – | |
| 2.3 | Cash flows from loans to other entities | – | – |
| 2.4 | Dividends received (see note 3) | – | – |
| 2.5 | Other (provide details if material) | – | – |
| 2.6 | Net cash from / (used in) investing activities | (117) | (117) |
| 3 | Cash flows from financing activities | ||
| 3.1 | Proceeds from issues of equity securities (excluding convertible debt securities) | – | – |
| 3.2 | Proceeds from issue of convertible debt securities | – | – |
| 3.3 | Proceeds from exercise of options | – | – |
| 3.4 |
Transaction costs related to issues of equity securities or convertible debt securities |
– | – |
| 3.5 | Proceeds from borrowings | – | – |
| 3.6 | Repayment of borrowings | (78) | (78) |
| 3.7 | Transaction costs related to loans and borrowings | – | – |
| 3.8 | Dividends paid | (1,976) | (1,976) |
| 3.9 | Other (provide details if material) | – | – |
| 3.10 | Net cash from / (used in) financing activities | (2,054) | (2,054) |
| 4 |
Net increase / (decrease) in cash and cash equivalents for the period |
||
| 4.1 | Cash and cash equivalents at beginning of period | 121,509 | 121,509 |
| 4.2 | Net cash from / (used in) operating activities (item 1.9 above) | 17,338 | 17,338 |
| 4.3 | Net cash from / (used in) investing activities (item 2.6 above) | (117) | (117) |
| 4.4 | Net cash from / (used in) financing activities (item 3.10 above) | (2,054) | (2,054) |
| 4.5 | Effect of movement in exchange rates on cash held | 972 | 972 |
| 4.6 | Cash and cash equivalents at end of period | 137,648 | 137,648 |
| 5 | Reconciliation of cash and cash equivalents at the end of the quarter (as shown in the consolidated statement of cash flows) to the related items in the accounts |
Current quarter $A’000 |
Previous quarter $A’000 |
|---|---|---|---|
| 5.1 | Bank balances | 34,857 | 27,066 |
| 5.2 | Call deposits | 102,450 | 94,100 |
| 5.3 | Bank overdrafts | – | – |
| 5.4 | Other (provide details) | 341 | 343 |
| 5.5 | Cash and cash equivalents at end of quarter (should equal item 4.6 above) | 137,648 | 121,509 |
| 6 | Payments to related parties of the entity and their associates | Current quarter $A’000 |
|---|---|---|
| 6.1 | Aggregate amount of payments to related parties and their associates included in item 1 | 2,648 |
| 6.2 | Aggregate amount of payments to related parties and their associates included in item 2 | |
| Note: if any amounts are shown in items 6.1 or 6.2, your quarterly activity report must include a description of, and an explanation for, such payments. | ||
| 7 | Financing facilities
Note: the term “facility” includes all forms of financing arrangements available to the entity. Add notes as necessary for an understanding of the sources of finance available to the entity. |
Total facility amount at quarter end $A’000 |
Amount drawn at quarter end $A’000 |
|---|---|---|---|
| 7.1 | Loan facilities | ||
| 7.2 | Credit standby arrangements | ||
| 7.3 | Other (please specify) | ||
| 7.4 | Total financing facilities | ||
| 7.5 | Unused financing facilities available at quarter end | ||
| 7.6 | Include in the box below a description of each facility above, including the lender, interest rate, maturity date and whether it is secured or unsecured. If any additional financing facilities have been entered into or are proposed to be entered into after quarter end, include a note providing details of those facilities as well. | ||
| 8 | Estimated cash available for future operating activities | $A’000 |
|---|---|---|
| 8.1 | Net cash from / (used in) operating activities (item 1.9) | 17,388 |
| 8.2 | Cash and cash equivalents at quarter end (item 4.6) | 137,648 |
| 8.3 | Unused finance facilities available at quarter end (item 7.5) | 0 |
| 8.4 | Total available funding (item 8.2 + item 8.3) | 137,648 |
| 8.5 | Estimated quarters of funding available (item 8.4 divided by item 8.1) | N/A |
| Note: if the entity has reported positive net operating cash flows in item 1.9, answer item 8.5 as “N/A”. Otherwise, a figure for the estimated quarters of funding available must be included in item 8.5. | ||
| 8.6 | If item 8.5 is less than 2 quarters, please provide answers to the following questions: | |
| 8.6.1 | Does the entity expect that it will continue to have the current level of net operating cash flows for the time being and, if not, why not? | |
| Answer: N/A | ||
| 8.6.2 | Has the entity taken any steps, or does it propose to take any steps, to raise further cash to fund its operations and, if so, what are those steps and how likely does it believe that they will be successful? | |
| Answer: N/A | ||
| 8.6.3 | Does the entity expect to be able to continue its operations and to meet its business objectives and, if so, on what basis? | |
| Answer: N/A | ||
| Note: where item 8.5 is less than 2 quarters, all of questions 8.6.1, 8.6.2 and 8.6.3 above must be answered. | ||
Compliance statement
- This statement has been prepared in accordance with accounting standards and policies which comply with Listing Rule 19.11A.
- This statement gives a true and fair view of the matters disclosed.
Date: 31 October 2022
Authorised by: MR DARREN KEAMY
(Name of body or officer authorising release – see note 4)
Notes
- This quarterly cash flow report and the accompanying activity report provide a basis for informing the market about the entity’s activities for the past quarter, how they have been financed and the effect this has had on its cash position. An entity that wishes to disclose additional information over and above the minimum required under the Listing Rules is encouraged to do so.
- If this quarterly cash flow report has been prepared in accordance with Australian Accounting Standards, the definitions in, and provisions of, AASB 107: Statement of Cash Flows apply to this report. If this quarterly cash flow report has been prepared in accordance with other accounting standards agreed by ASX pursuant to Listing Rule 19.11A, the corresponding equivalent standard applies to this report.
- Dividends received may be classified either as cash flows from operating activities or cash flows from investing activities, depending on the accounting policy of the entity.
- If this report has been authorised for release to the market by your board of directors, you can insert here: “By the board”. If it has been authorised for release to the market by a committee of your board of directors, you can insert here: “By the [name of board committee – eg Audit and Risk Committee]”. If it has been authorised for release to the market by a disclosure committee, you can insert here: “By the Disclosure Committee”.
- If this report has been authorised for release to the market by your board of directors and you wish to hold yourself out as complying with recommendation 4.2 of the ASX Corporate Governance Council’s Corporate Governance Principles and Recommendations, the board should have received a declaration from its CEO and CFO that, in their opinion, the financial records of the entity have been properly maintained, that this report complies with the appropriate accounting standards and gives a true and fair view of the cash flows of the entity, and that their opinion has been formed on the basis of a sound system of risk management and internal control which is operating effectively.